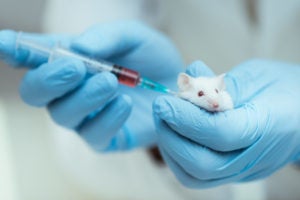
SEOUL—Humane Society International/Korea is calling on legislators to back a series of tough new legal measures to address the lack of progress by Korea’s relevant regulatory and research funding ministries toward reduction and replacement of animal testing. Statistics on laboratory animal use in 2019 published this week by Korea’s Ministry of Agriculture, Food and Rural Affairs’ Animal and Plant Quarantine Agency[1] revealed a marginal 0.4% decrease in total animal use compared to the previous year (3,712,380 in 2019 compared to 3,727,163 in 2018), together with alarming increases in animal use for testing insecticides (+187%), industrial chemicals (+115%), education and training (+77.8%), pharmaceutical quality control testing (+40%), production of genetically modified animals (+12%), and experiments in the most severe pain category (+9.7%).
Borami Seo, HSI/Korea senior policy manager for research and toxicology, said: “These statistics make it clear far Korean authorities and science funding ministries have to go to fulfill their stated commitment to replace, reduce or even minimize the most severe suffering in animal testing. The future lies in human mimetic non-animal approaches like human organoids, organs-on-chips and next-generation computing and AI, not in poisoning or genetically modifying mice, monkeys and other animals. It’s time our government followed the example of the United States, the Netherlands and other innovation economies by making a serious investment in non-animal technologies to advance safety science and medical research.”
HSI/Korea has been working with members of the National Assembly and key ministries to make research and regulatory testing with non-animal approaches a higher priority. For example, Korean chemical, pesticide and pharmaceutical authorities should reflect on their performance in comparison to the US Environmental Protection Agency’s commitment to reduce mammalian testing requirements by 30% by 2025 and to completely eliminate them by 2035. Korean science funding ministries should look to European and American funding programs for organ-on-a-chip technologies to advance drug testing and human disease research.
Assembly member In-soon Nam and HSI will host an Assembly forum on June 30 to discuss a new legislative initiative to promote the development, distribution, and use of alternatives to animal testing methods. Presenters include officials from the Ministry of Food and Drug Safety and the Ministry of Health and Welfare, the president of the Korea Institute of Toxicology Chang-Woo Song, the vice president of the Korean Society for Alternative to Animal Experiments Gwang-Man Kim, CEO of Dana Green Bio Ki-woo Kim, and HSI/Korea. Additionally, legal experts from the National Assembly’s Legislative office, the Korea Legislation Research Institute and the Korean lawyers’ group People for Non-Human Rights will join the discussion.
END
Media contact: Borami Seo, bseo@hsi.org
- MAFRA’s full statistical report for 2019 is available online (in Korean).